24 hour contact: 01707 666297
Laminitis Research
Laminitis is a common and painful condition of the adult equine, often resulting in permanent lameness or euthanasia.
Reported estimates of laminitis frequency range from 1.5-34%1, depending on the population studied (general practice or referral institutions), proportion of ponies versus horses, presence of intercurrent diseases (particularly gastrointestinal disease) and the geographic location (generally USA, UK or Australia). It may occur as a single episode or, more commonly, as repeated bouts over a prolonged period (recurrent laminitis). The risk of mortality was increased nearly 6-fold by the presence of laminitis in a population of horses treated in first opinion practices in the UK indicating the importance of this disease1.
Laminitis is now considered to be a clinical syndrome associated with systemic disease (sepsis or systemic inflammatory response syndrome [SIRS] or endocrine disease) or altered weight bearing rather than being a discrete disease entity. Thus, laminitis can be divided into three forms, sepsis-associated, endocrinopathic and supporting limb laminitis.

What we have learnt from Previous Research at RVC
Research into laminitis at the Royal Veterinary College (RVC) has focussed on endocrinopathic laminitis. Endocrinopathic laminitis encompasses laminitis that occurs in animals with an underlying endocrine disease that results in insulin dysregulation (ID) i.e. equine metabolic syndrome (EMS) and pituitary pars intermedia dysfunction (PPID; formerly Equine Cushing’s disease). EMS is a cluster of clinical abnormalities associated with an increased risk of laminitis. The key feature of EMS is ID. Additional features include obesity, high circulating triglyceride concentrations and adipose tissue dysregulation which results in altered production of adipose-tissue derived hormone (adipokines) such as adiponectin and leptin. PPID is a progressive neurodegenerative disorder associated with loss of the inhibitory dopaminergic input to the pituitary pars intermedia (PI). This results in increased production of the normal hormone products of the PI, some of which may antagonise the actions of insulin resulting in ID in a subset of animals. However, not all animals with ID develop laminitis. There appear to be genetic and environmental influences as well.
Over the last 10 years, through research conducted at the RVC we have improved knowledge in a range of areas:
1) Equine metabolic syndrome (EMS)
We have shown that grazing summer pasture2 or the consumption of dietary carbohydrate3 appears to induce metabolic changes including the worsening of ID and the development of hypertension.
ID in animals with EMS manifests in one of three forms, namely resting (basal) hyperinsuilnaemia, an excess insulin response to oral carbohydrate and tissue insulin resistance. The excess insulin response to oral carbohydrate can be identified by feeding the animal either a small amount of glucose powder or corn syrup. The results from these two tests, whilst not identical, agreed in the majority (85%) of animals4. An improved dose of corn syrup to identify ID has also been determined5 as well as the effect of withholding food prior to the test6.
There are a number of assays that are used to quantify insulin concentrations in equine serum. Agreement among results of the different assays was variable and the results cannot be used interchangeably7.
Animals with EMS had a reduced anti-inflammatory capacity rather than a chronic pro-inflammatory state8; this was improved by 14 days of regular low intensity (walking and trotting) exercise9.
Apart from seasonal hypertension2, other methods did not demonstrate evidence of endothelial dysfunction in ponies with EMS10.
Circulating concentrations of the insulin-sensitising and anti-inflammatory adipose-tissue derived hormone adiponectin were lower in animals that had previously had laminitis8 and also in non laminitic animals that developed laminitis in the next 1, 2 or 3 years11. Thus, hypoadiponectinaemia is a risk factor for future laminitis. An immunoturbidimetric assay was validated for measurement of adiponectin in equine samples12.
2) Treatment of laminitis
Evaluation of cases of endocrinopathic laminitis treated by first opinion veterinarians revealed that eight weeks after the onset of the laminitis, 95% of animals were still alive and 59% were being ridden again13. Survival was associated with a lower body weight, optimal body condition and mild rather than severe laminitis.13 Being ridden again, was more likely in animals without previous laminitis. No one medical therapy or foot support methods was superior to any other13.
3) Laminitis epidemiology
Over a 6-year period, the highest prevalence and incidence of laminitis occurred in May; there was a positive association between the hours of sunshine and the prevalence and incidence of laminitis; and females and lighter animals had the greatest risk of developing laminitis14.
4) Diagnosis of pituitary pars intermedia dysfunction (PPID)

A diagnosis of PPID is made by measuring plasma adrenocorticophic hormone (ACTH) concentrations. There are two assays that are validated for measurement of ACTH in equine samples. The two methods yielded different absolute values and it would appear that other pituitary-derived hormones interfere with the chemiluminescent-immunoassay15.
5) Management of obesity
Obesity is an additional feature of EMS and some animals with PPID are overweight/obese. Management strategies are employed to reduce weight in these animals which often include geographic restriction of pasture. Accelerometers were validated for activity tracking in horses and used to determine the effect of small vs. standard paddock size of time spent grazing and locomoting16. We are also investigating the effects of different types of strip grazing on physical activity in ponies.
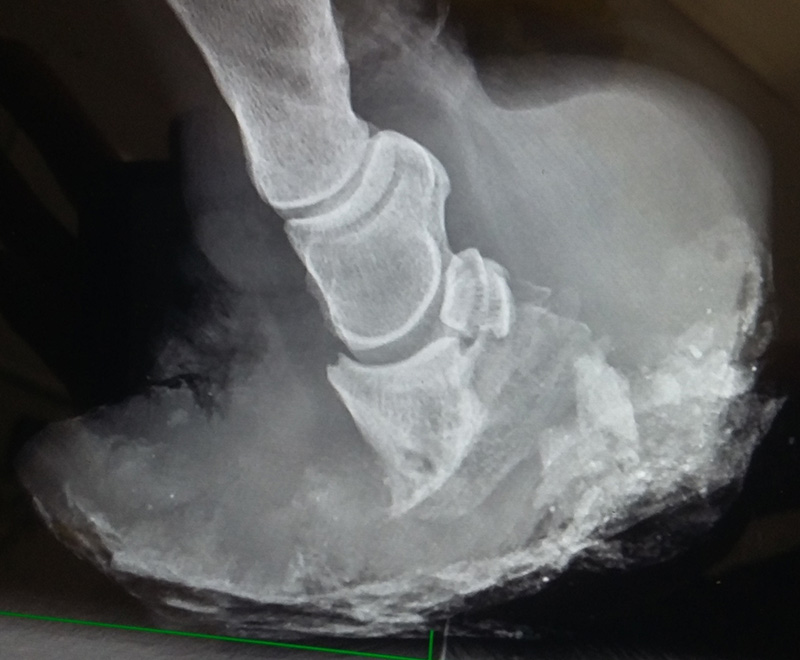
6) The mechano-biology of hoof growth
In order to understand how diseases may affect hoof growth, we first need to understand how the normal hoof growth. Combination of maths, physics and histological examination of hooves revealed how the synthesis of the hoof capsule starts from the coronet and the soft papillae undergo gradual transition into hard keratinised tissue.17 In addition, it revealed how it is possible for the hoof to grow at a rate that is faster than the potential proliferation rate of epithelial cells and how the hoof can grow in a curved manner rather than the usual straight manner as a result of faster growth from the coronary band at the quarters compared to the toe region17.
Current research at the RVC
There are three projects currently on going at the RVC. The first is a cohort study in which a group of 250 ponies that have never had laminitis before are being visited every 6 months for 4 years. Phenotypic, metabolic and management data is collected at each visit. At the end of the study, the ponies that have developed laminitis for the first time during the study period will be compared to those that have remained non-laminitic to determine the differences between them.
The second study is focusing on horse carers’ awareness of evidence-based best practices and to identify barriers and drivers affecting the implementation of research-based recommendations with respect to laminitis prevention. An online survey was completed by 1,414 respondents who answered questions relating to a wide range of topics including where they obtained information relating to lameness, weight management and grazing, their thoughts on scientific research and whether they were able to practice management techniques known to reduce the risk of laminitis. The results obtained are currently being analysed. The next part of the study will involve conducting interviews with some of the respondents in order to explore this further with a small number of selected horse carers including horse owners, farriers and equine vets.
The final study is to further elucidate the role of the adipose tissue derived hormone adiponectin in endocrinopathic laminitis. Firstly, this study will determine whether adiponectin receptors are present within the equine lamellae, will determine whether there is crosstalk between the adiponectin, insulin and insulin-like growth factor-1 signalling pathways. Secondly, the effect of hyperinsulinaemia, induction of tissue insulin resistance and pasture-induced weight gain on circulating adiponectin concentrations will be investigated in vivo. Finally, in human metabolic syndrome, dietary manipulation and pharmacologic agents are used to increase circulating adiponectin concentrations and in turn reduce the associated cardiovascular disease risk. This project will determine whether similar approaches can be used in EMS. The effect of weight loss with or without dietary supplementation on circulating adiponectin concentrations will be evaluated. Potential pharmacologic agents will be screened in vitro through evaluation of their effects on adipocyte adiponectin production.
References
- Welsh CE, Duz M, Parkin TDH, et al. Disease and pharmacologic risk factors for first and subsequent episodes of equine laminitis: A cohort study of free-text electronic medical records. Prev Vet Med 2017;136:11-18.
- Bailey SR, Habershon-Butcher J, Ransom K, et al. Hypertension and insulin resistance in a mixed-breed population of ponies predisposed to laminitis: Further characterizing pre-laminitic metabolic syndrome. Am J Vet Res 2008;69:122-129.
- Bailey SR, Menzies-Gow NJ, Harris PA, et al. Effect of dietary fructans and dexamethasone administration on the insulin response of ponies predisposed to laminitis. J Am Vet Med Assoc 2007;231:1365-1373.
- Smith S, Harris PA, Menzies-Gow NJ. Comparison of the in-feed glucose test and the oral sugar test. Equine Vet J 2016;48:224-227.
- Jocelyn NA, Harris PA, Menzies-Gow NJ. Effect of varying the dose of corn syrup on the insulin and glucose response to the oral sugar test. Equine Vet J 2018.
- Knowles EJ, Harris PA, Elliott J, et al. Use of the oral sugar test in ponies when performed with or without prior fasting. Equine Vet J 2017;49:519-524.
- Knowles EJ, Harris PA, Elliott J, et al. Insulin assay choice affects results of the oral sugar test. Dorothy Russell Havemeyer Equine Endocrinology Symposium 2017;39.
- Wray H, Elliott J, Bailey SR, et al. Plasma concentrations of inflammatory markers in previously laminitic ponies. Equine Vet J 2013;45:546-551.
- Menzies-Gow NJ, Wray H, Bailey SR, et al. The effect of exercise on plasma concentrations of inflammatory markers in normal and previously laminitic ponies. Equine Vet J 2013.
- Finding ET. Does endothelial cell dysfunction underlie the predisposition to pasture-associated equine laminitis?: Univerity of London, 2017.
- Menzies-Gow NJ, Harris PA, Elliott J. Prospective cohort study evaluating risk factors for the development of pasture-associated laminitis in the United Kingdom. Equine Vet J 2017;49:300-306.
- Menzies-Gow NJ, Knowles EJ, Rogers I, et al. Validity and application of immunoturbidimetric and enzyme-linked immunosorbent assays for the measurement of adiponectin concentration in ponies. Equine Vet J 2018.
- Menzies-Gow NJ, Stevens K, Barr A, et al. Severity and outcome of equine pasture-associated laminitis managed in first opinion practice in the UK. Vet Rec 2010;167:364-369.
- Menzies-Gow NJ, Katz LM, Barker KJ, et al. An epidemiological study of pasture-associated laminitis and concurrent risk factors in the South of England. Vet Rec 2010.
- Knowles EJ, Moreton-Clack MC, Shaw S, et al. Plasma adrenocorticotropic hormone (ACTH) concentrations in ponies measured by two different assays suggests seasonal cross-reactivity or interference. Equine Vet J 2018;50:672-677.
- Maisonpierre IN, Sutton MA, Harris P, et al. Accelerometer activity tracking in horses and the effect of pasture management on time budget. Equine Vet J 2019.
- Al-Agele R, Paul E, Taylor S, et al. Physics of animal health: on the mechano-biology of hoof growth and form. J R Soc Interface 2019;16:20190214.


