The regulation of soft tissue and skeletal calcification
Physiological bone formation is essential for the normal functioning of the skeleton. In contrast, calcification within soft tissues has numerous negative physiological effects. Consequently, there are multiple mechanisms in place to prevent soft tissues from calcifying. When these regulatory processes fail unwanted mineral deposition can occur, a good example being vascular calcification. Vascular calcification is the formation of bone-like deposits within blood vessels and heart valves and is a significant risk factor for future adverse cardiovascular events such as heart attack and stroke. Despite recent advances in understanding about how vascular calcification develops, there are still no effective treatments to prevent or treat this condition.
Earlier research has shown that there are some outward similarities between bone formation and vascular calcification. Using the expertise at the RVC in both skeletal and cardiovascular biology we have been using a combination of cell-based in vitro experiments and rodent models to fully understand the similarities and differences between these two processes. Using human and rodent vascular and bone cells we have identified several compounds that inhibit vascular calcification but promote bone formation in vitro. These compounds of interest are then investigated in a rat model of vascular calcification (induced by feeding a modified diet) to determine if they have beneficial effects in a whole-body setting. All animals are carefully monitored to ensure that they are not suffering because of the modified diet or pharmacological interventions.
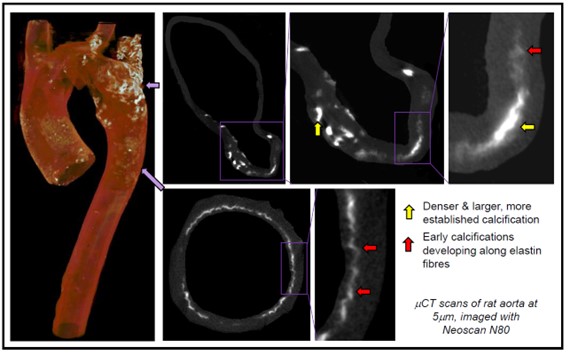
A recent BBSRC equipment grant (BB/Z51547/1) has allowed the RVC to purchase a state-of-the-art x-ray microcomputed tomography (mCT) scanner (Neoscan N80). mCT imaging allows the bone and vasculature to be visualised in high resolution and generates detailed, quantitative 3D data. Exploiting the technical advantages of this new system, we have recently reanalysed archived samples from earlier rodent studies. These analyses have revealed novel and important information about the location, size and pattern of vascular calcification development without using further experimental animals.
Going forward and continuing in our drive to reduce and replace the use of animals in research we have recently embarked on a project to completely humanise the in vitro and ex vivo models of vascular calcification. This research, funded by The Humane Research Trust, will develop novel protocols to study vascular calcification that do not use any animals or their derived products.
Ultimately, the overarching aim of this research is to identify therapies which can be used to treat vascular calcification without causing bone loss.