
The role of cell senescence in the age-related dysregulation of protein turnover in tendon
The study will allow us to understand, for the first time, the link between cell senescence and protein turnover in tendon. By establishing the effect of reversing tendon cell senescence on protein synthesis and breakdown, we may discover new therapies that can be used to prevent and treat age-related tendon injuries in the future, both in humans and horses.
Challenge
Tendons connect muscles to bone and are essential for movement. They stretch and recoil as we walk and run, improving the efficiency of our movements. Even taking a few steps without the Achilles tendon connecting our calf muscle to our heel would be very difficult. Unfortunately tendons are regularly injured, not only in athletes, but also during normal every day activities, particularly as we get older. Injuries are painful and can be highly limiting, preventing usual activity and decreasing quality of life. They cost the economy millions of pounds every year, not only in treatment costs, but also due to the indirect costs of both work absence and reduced health due to an inability to exercise. Horses also often suffer from tendon injuries, and provide an excellent model in which to study tendon ageing. The risk and characteristics of tendon injury are very similar between humans and horses, and tendons can easily be obtained from young and old horses that are put to sleep for reasons other than tendon injury.
Tendon injuries in both humans and horses are difficult to treat due to a poor understanding of the underlying causes of the age-related decrease in tendon function that leads to increased risk of injury. In other tissues within the body, a common cause of age-related diseases is disruption of the balance between the synthesis and breakdown of proteins that make up the tissue. This process is known as proteostasis, and must be carefully regulated to maintain tissue health. As a tissue ages, its resident cells often lose their ability to grow and divide, and also begin to produce molecules than can cause protein breakdown. These cells are referred to as senescent cells, and cell senescence can disrupt the balance between protein synthesis and breakdown within a tissue. This can reduce the tissue's ability to function properly, which may eventually lead to injury.
Solution
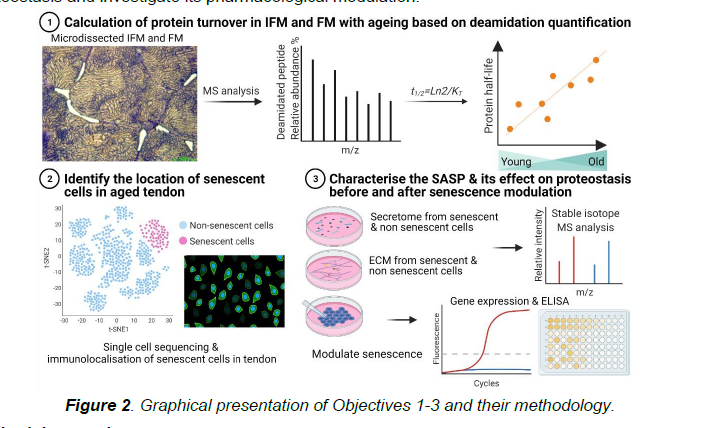
In this study, we are investigating the relationship between proteostasis and cell senescence in tendon with ageing. First, we will establish how proteostasis alters with ageing in different regions of the horse forelimb superficial digital flexor tendon (SDFT). We will then identify the location of the senescent cells within the SDFT from aged horses, and develop methods to isolate these cells from tendons so we can study them in the laboratory. Using these isolated cells, we will establish how protein synthesis and breakdown changes when the cells become senescent. Finally, we will determine if reversal of senescence by treating the cells with small molecules can restore the balance between protein synthesis and breakdown.
Impact
By establishing the effect of reversing tendon cell senescence on protein synthesis and breakdown, we may discover new therapies that can be used to prevent and treat age-related tendon injuries in the future, both in humans and horses.
Partners
Funded by BBSRC
Richard Faragher, University of Brighton
Publications
| Title | Publication | Year |
| The Interfascicular Matrix of Energy Storing Tendons Houses Heterogenous Cell Populations Disproportionately Affected by Aging | Aging and Disease | 2024 |
