
Erythrocyte invasion in malaria: molecular mechanism to precision therapeutics
Researchers at the RVC and Oxford University are leading an innovative research project funded by the Wellcome Trust to determine the structure and function of a parasite-derived protein complex resulting in malaria progression.
Challenge
Malaria is a deadly disease caused by Plasmodium parasites. Out of the six species which infect humans, Plasmodium falciparum causes the most severe disease outcomes. Each year, over 200 million cases and ca.600,000 deaths occur world-wide, disproportionately affecting young children in Africa. With these numbers expected to rise over the coming years due to growing parasite resistance to frontline anti-malarial drugs, the development of highly effective malaria vaccines is desperately needed.
Disease symptoms arise from continuous cycles of parasites invading red blood cells (RBCs), replicating within them, and then bursting free to infect new RBCs. Invasion is a rapid, multi-step process, during which the parasite secretes specialised RBC-binding proteins to facilitate entry into its new RBC home. Importantly, disrupting these molecular interactions between parasite and host cell prevents invasion and thus, halts disease progression. Therefore, parasite proteins required for invasion are good therapeutic targets. Understanding the detailed molecular functions of parasite proteins involved in invasion is critical therefore to rationally design vaccines as well as malaria therapies.
Solution
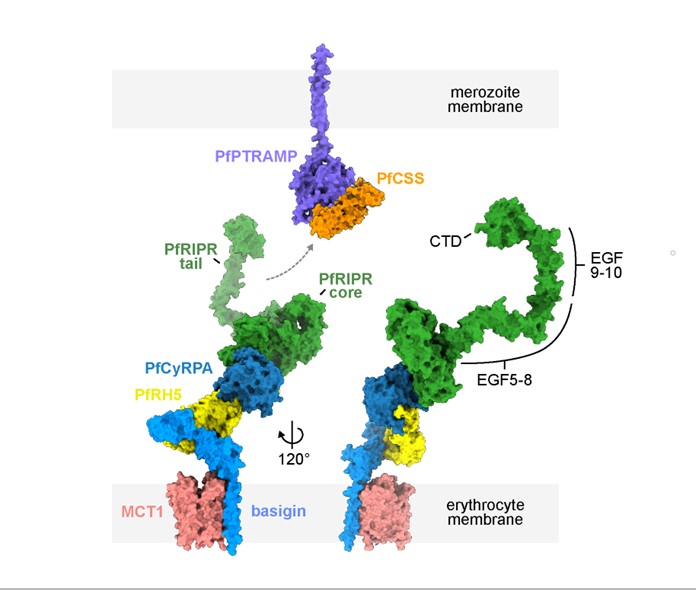
One essential invasion ligand and leading vaccine candidate is the P. falciparum ‘reticulocyte binding protein homolog 5’ (PfRh5), which is part of a five-member protein complex assembled by the parasite just prior to invasion in specialised secretory organelles. During invasion, PfRh5 binds to basigin receptor on the RBC surface. However, little else is understood about the mechanisms by which this essential protein complex acts during invasion. Gaining insight into the specific functions of this important complex will guide the design of precision therapeutics to block invasion.
Working with the Higgins lab at the University of Oxford ( https://higginslab.web.ox.ac.uk/), our approach will entail using structure-guided molecular approaches to investigate how the PfRh5 protein complex is assembled, activated and how it functions as a whole, as well as which components are vulnerable to invasion neutralising antibodies. For this we will be combining protein biochemistry, structural biology, molecular and cellular biological approaches and test our findings on in vitro Plasmodium tissue culture models.
Impact
In the past two years a breakthrough on malaria vaccines has been achieved, and although this is a huge step forward in the fight against malaria, the newly approved vaccines do not fully protect each child. We therefore urgently need further vaccines and other therapeutics to control malaria and to eradicate it in the future.
RBC invasion by the malaria parasite is associated with the clinical symptoms of malaria. Proteins involved in this invasion process are good vaccine targets we can exploit. Previous studies identified the receptor-binding domain of PfRh5 as one of those promising vaccine targets. Little is known about other protein domains in the PfRh5-led multimeric protein complex and their role in RBC invasion or susceptibility to neutralising antibodies.
Here we will be determining the structure of this protein complex as well as study the function of individual complex members during RBC invasion to facilitate rational vaccine and therapeutic antibody design to tackle this deadly disease.
Partners
Higgins lab, at the University of Oxford ( https://higginslab.web.ox.ac.uk/). Wellcome Trust ( https://wellcome.org/).
