Dr Bradley Cobb
Department: Comparative Biomedical Sciences
Campus: Camden
Research Groups: Comparative Genomics, Endocrinology and Reproduction, CPCS (Research Programme)
Bradley is a Senior Lecturer in Comparative Biomedical Sciences. His main research focus is on the regulation of gene expression in development.
Bradley joined the staff at the RVC as a Senior Lecturer in September 2008. He received a PhD in Microbiology from the University of Virginia and graduated from the University of California, Santa Barbara with a BA in Aquatic Biology followed by an MA in Molecular Biology. He did postdoctoral research at the University of Chicago and the University of California, Los Angeles. In addition, he has also held positions as a Research Assistant Professor at the University of California, Los Angeles and a Senior Research Scientist at the MRC Clinical Sciences Centre within the Imperial College School of Medicine, London.
Bradley’s overall interest is in the mechanisms regulating gene expression during development. The primary interest is in the roles of microRNAs (miRNAs), which are small RNAs (≈22 mers) that post-transcriptionally regulate gene expression by binding to target sequences in messages and inhibiting their translation and stability. Gene regulation by miRNAs is critical at many developmental stages in organisms ranging from worms to man, and Bradley’s research has examined the functions of miRNAs in lymphocyte and early embryonic development.
In previous work, Bradley analyzed miRNA function in T cell development by creating and characterizing a conditional knock out of the Dicer gene, which encodes an RNase required for miRNA synthesis. Loss of Dicer resulted in a depletion of regulatory T (Treg) cells, which are required for down regulation of the immune response and the prevention of autoimmunity. Recent work has identified critical miRNAs and their regulation of important genes that modulate Treg development. In addition, work in the lab has identified that miRNAs also play a key role in regulating the development of another subset of helper T cells called Th9. These are important in many allergic responses but also mediate anti-tumor responses in many cancers. Therefore, the current interest of Bradley’s research is how miRNAs regulate expression of key genes that determine the differentiation of helper T cells into the various subsets. Deciphering such mechanisms will be essential to understand the regulatory pathways involved in mediating an appropriate immune response, which eliminates pathogens and tumor cells but does not attack normal host tissue and there by lead to autoimmunity.
Further research in Bradley’s laboratory has examined the roles of miRNAs in the early embryo. Murine embryonic stem cells (ES) cells have been used as a model system to study miRNA function, and Dicer deleted ES cells have been found to have a defect in calcium homeostasis between the endoplasmic reticulum (where most calcium is stored inside of the cell) and the cytoplasm, indicating that miRNAs regulate genes involved in the transport of calcium between these two sites. Since calcium signaling and miRNAs play an important role in the differentiation of the early embryo, current work is in understanding how these two are interrelated. This research involves determining the critical genes regulated by miRNAs that control calcium homeostasis and how this mediates key steps in the differentiation of the early embryo.
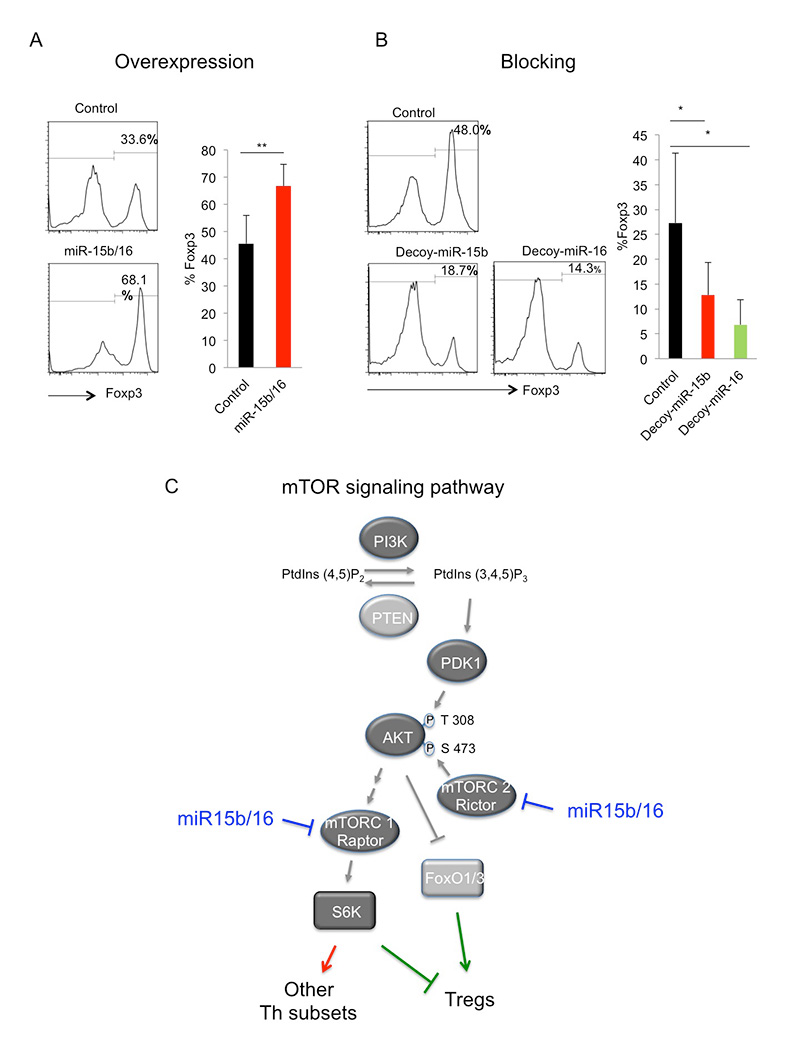
Fig. 1. The miRNAs miR-15b/16 enhance induction of Tregs by regulating the expression of the mTOR signaling components Rictor and mTOR. A. Overexpression of miR-15b/16 enhances the induction of Tregs as measured by FACS staining of the Treg specific transcription factor Foxp3. B. Blocking of miR-15b/16 through overexpression of a decoy target site (acting as a competitive inhibitor of miR-15b/16) inhibits Treg induction. C. A simplified diagram of the mTOR signaling pathway whose suppression leads to the induction of Tregs. miR-15b/16 suppresses the expression of the components Rictor and mTOR, and thus inhibits signaling, which leads to increased Treg induction.
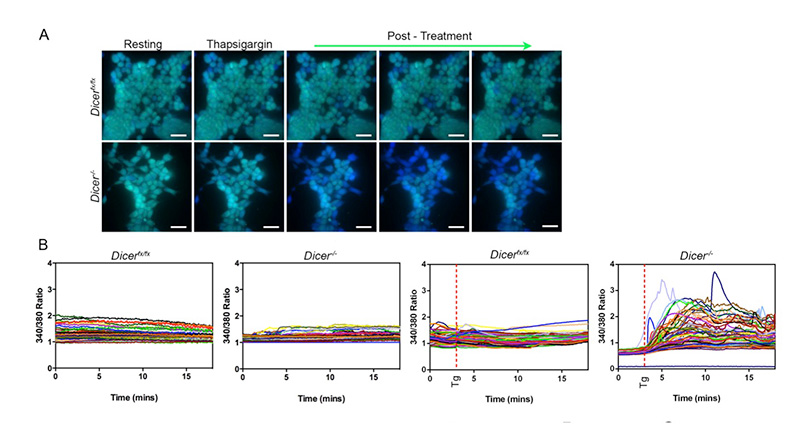
Fig. 2. ES cells lacking Dicer are defective at maintaining calcium homeostasis upon treatment with thapsigargin, which inhibits calcium uptake into the endoplasmic reticulum. Wild type (Dicerfx/fx) ES cells and those lacking Dicer (Dicer-/-) were loaded with the calcium dye Fura2, and cytoplasmic calcium levels were analyzed by fluorescence microscopy every five seconds over 18 minutes at both 340 and 380 nm. After three minutes, cells were either left untreated or were treated with thapsigargin (Tg). (A) Representative images from a single experiment are shown of cells treated with thapsigargin. More Dicer-/- cells responded to thapsigargin with increased cytoplasmic calcium levels (blue cells). Scale bars represent approximately 25 ?m. (B) Representative graphs of untreated or thapsigargin treated cells, plotting the ratio between 340 and 380 nm of every cell in a field of view also demonstrate that many more Dicer-/- ES cells respond to thapsigargin treatment with increased cytoplasmic calcium levels.
Selected Publications
1. Singh Y, Garden O, Lang F, Cobb BS. 2016. miRNAs regulate T cell production of IL-9 and identify hypoxia inducible factor-2? as an important regulator of Th9 and Treg development. Immunology. 149:74–86.
2. Singh Y, Garden O, Lang F, Cobb BS. 2015. MicroRNA-15b/16 enhances the induction of regulatory T cells by regulating the expression of Rictor and mTOR. J Immunol. 195:5667-77. (Featured with a lay summary in the online journal Atlas of Science http://atlasofscience.org/how-the-immune-system-is-regulated-by-small-rnas-called-micrornas-to-prevent-self-attacks-leading-to-autoimmunity/)
3. Pernaute B, Spruce T, Smith KM, Sanchez Nieto JM, Manzanares M, Cobb BS, Rodriguez T. 2014. MicroRNAs control the apoptotic threshold in primed pluripotent stem cells through regulation of BIM. Genes & Devel. 28:1873-78.
4. Spruce T, Pernaute B, Di-Gregorio A, Cobb BS, Merkenschlager M, Manzanares M, Rodriguez TA. 2010. An early developmental role for miRNAs in the maintenance of extraembryonic stem cells in the mouse embryo. Dev Cell. 19:207-19.
5. Taylor B, Cobb BS, Bruno L, Webster Z, Fisher AG, Merkenschlager M. 2009 A reappraisal of evidence for probabilistic models of allelic exclusion. PNAS. 106:516-21.
6. Koralov SB, Muljo SA, Galler GR, Krek A, Chakraborty T, Kanellopoulou C, Jensen K, Cobb BS, Merkenschlager M, Rajewsky N, and K Rajewsky. 2008. Dicer ablation affects antibody diversity and cell survival in the B lymphocyte lineage. Cell. 132:860-74.
7. Thompson EC, Cobb BS, Sabbattini P, Meixlsperger S, Parelho V, Liberg D, Taylor B, Dillon N, Georgopoulos K, Jumaa H, Smale ST, Fisher AG, and M Merkenschlager. 2007. Ikaros DNA binding proteins as integral components of developmental stage-specific regulatory circuits. Immunity. 26:335-344.
8. Cobb BS, Hertweck A, Smith J, O’Connor E, Graf D, Cook T, Smale ST, Sakaguchi S, Livesey FJ, Fisher AG, and M Merkenschlager. 2006. A role for Dicer in immune regulation. J Exp Med. 203:2519-27.
9. Cobb BS, Nesterova TB, Thompson E, Hertweck A, O'connor E, Godwin J, Wilson CB, Brockdorff N, Fisher AG, Smale ST, and M Merkenschlager. 2005. T cell lineage choice and differentiation in the absence of the RNase III enzyme Dicer. J Exp Med. 9:1367-73.
10. Cobb BS and ST Smale. 2005. Ikaros-family proteins: in search of molecular functions during lymphocyte development. Curr Top Microbiol Immunol. 290:29-47.
Bradley teaches Biochemestry, Molecular Biology, and Immunology.
